
Homemade Hydrogen | EXPERIMENT | Aluminium foil & Caustic soda | Crazy Reaction | KesPra ✓ - YouTube

When metal X is treated with sodium hydroxide, a white precipitate (A) is obtained, which is soluble in excess of NaOH to give soluble complex (B) .Compound (A) is soluble in dilute

Caustic soda vs Aluminium foil || Caustic soda and Aluminium foil reaction || Caustic soda reaction - YouTube

Doubt: Sir in this question why we use combine gas equation this equation only applicable on gas but here they given bits of aluminum ??? Chapter: States of Matter - Subject: Chemistry -

Mixing Caustic Soda + Aluminium Foil to produce Hydrogen Gas | Science Experiment | Dear.hacker - YouTube
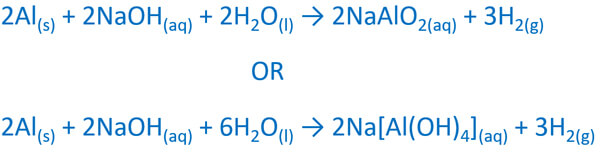
i) State with balanced equation, what happens when (a) Aluminium is reacted with hot conc caustic soda solution. - Sarthaks eConnect | Largest Online Education Community

The drain cleaner Drainex contains small bits of aluminium which reacts with caustic soda to produce dihydrogen gas. What volume of dihydrogen at 20°C and 1 bar will be released when 0.15
The drain cleaner, Drainex contains small bits of Aluminium which react with caustic soda to produce dihydrogen. What volume of dihydrogen at 20 degree C and one bar will be released when

what will you observe when freshly precipitated aluminium hydroxide reacts with caustic soda solution - Brainly.in

Name the gas in each of the following:The gas evolved on reaction of aluminium with boiling concentrated caustic alkali solution












![What happens when?Aluminium is heated with caustic soda solution. [I.I.T. 1997 ] What happens when?Aluminium is heated with caustic soda solution. [I.I.T. 1997 ]](https://haygot.s3.amazonaws.com/questions/1676274_1728047_ans_b94d71e267324c9cb872fa6b57978e72.jpg)


